Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (49): 7948-7954.doi: 10.3969/j.issn.2095-4344.2014.49.014
Previous Articles Next Articles
Variation of T cell subset during acute rejection after liver transplantation in rhesus monkeys
Ran Jiang-hua, Liu Jing, Zhang Xi-bing, Zhang Sheng-ning, Wu Shu-yuan, Li Lai-bang, Li Wang, Li Li
- Department of Hepatobiliary and Pancreatic Surgery, the Affiliated Ganmei Hospital of Kunming Medical University, Liver Transplantation Center of Organ Transplantation Institute of Yunnan Province, Kunming 650011, Yunnan Province, China
-
Revised:2014-09-15Online:2014-11-30Published:2014-11-30 -
Contact:Li Li, M.D., Professor, Doctoral supervisor, Department of Hepatobiliary and Pancreatic Surgery, the Affiliated Ganmei Hospital of Kunming Medical University, Liver Transplantation Center of Organ Transplantation Institute of Yunnan Province, Kunming 650011, Yunnan Province, China -
About author:Ran Jiang-hua, M.D., Professor, Master’s supervisor, Department of Hepatobiliary and Pancreatic Surgery, the Affiliated Ganmei Hospital of Kunming Medical University, Liver Transplantation Center of Organ Transplantation Institute of Yunnan Province, Kunming 650011, Yunnan Province, China -
Supported by:the Major Program of Kunming Science and Technology Bureau, No. 08S100304
CLC Number:
Cite this article
Ran Jiang-hua, Liu Jing, Zhang Xi-bing, Zhang Sheng-ning, Wu Shu-yuan, Li Lai-bang, Li Wang, Li Li. Variation of T cell subset during acute rejection after liver transplantation in rhesus monkeys[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(49): 7948-7954.
share this article
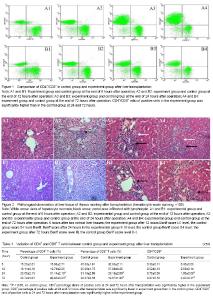
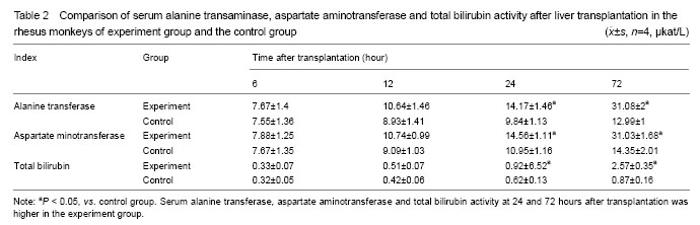
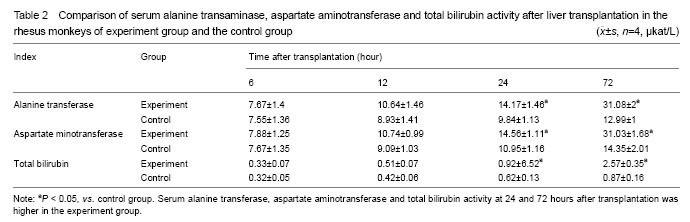
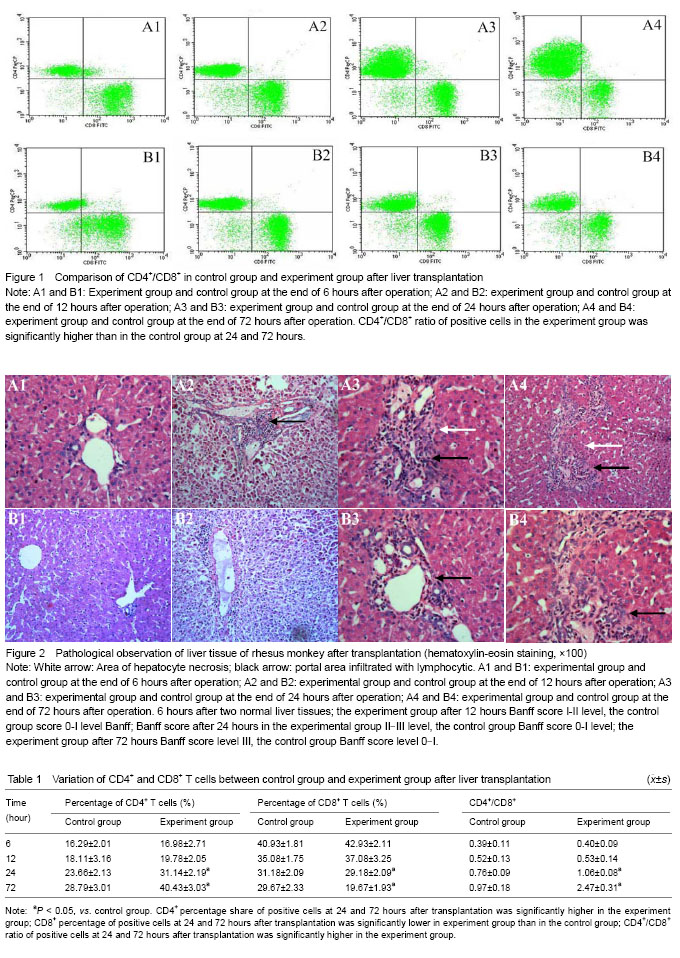
Variation of T-lymphocyte subsets In the experiment group without treatment of immunosuppressant, the amounts of CD4+ and CD8+ cells were significantly more than that of control group on the scatter diagram (Figure 1). Regarding the amount of CD4+ and CD8+ cells and the ratio of CD4+/CD8+, there was no significant differences between two groups (P > 0.05) at 6 and 12 hours, but the significant differences were found at 24 and 72 hours (P < 0.05). With the extension of time, the variation trend of CD4+ T cell amount in peripheral blood of experiment group and control group since 6 hours to 72 hours after operation was as follows: CD4+ T cell amount in peripheral blood of experiment group and control group increased with the extension of time; the increasing extent of experiment group was far greater than that of control group; CD8+ T cells amount in peripheral blood of experiment group and control group presented a decreasing trend with the extension of time, the decreasing extent of experiment group was significantly superior to that of control group (Table 1). Variation of biochemical indices of liver damage Comparison of serum alanine transaminase, aspartate aminotransferase and total bilirubin of rhesus monkeys in two groups after liver transplantation: alanine transaminase, aspartate aminotransferase and total bilirubin of experiment group were significantly higher than that of control group at 24 and 72 hours after operation (P < 0.05). The animals in experiment group did not take anti-rejection drugs or hormone after operation, so the damage of hepatic cells caused by the attack of inflammatory cells was more severe than that of control group. Alanine transaminase, aspartate aminotransferase and total bilirubin of two groups presented increasing trend with the extension of time. These three detection indices of two groups were close at 6 and 12 hours after operation, and the differences between two groups had no statistical significance (P > 0.05). Hepatic function presented increasing trend with the extension of time after liver transplantation, the differences of the interior-group in two groups had statistical significance (P > 0.05). Furthermore, the variation of experiment group was greater than of control group, especially from 24 to 72 hours after operation (Table 2). Histopathology of rhesus monkey hepatic tissue after liver transplantation Hepatic tissue of experiment group and control group was basically normal at 6 hours after operation, and no rejection feature was found under microscope (Figure 5, A1 and B1). At the end of 12 hours after operation, hematoxylin-eosin staining indicated the hepatic tissue of experiment group had a mild rejection, Banff score was I-II; the rejection grade of control group was undefined, Banff score was 0-I (Figure 2, A2 and B2). At the end of 24 hours after operation, hematoxylin-eosin staining indicated the hepatic tissue of experiment group had a mediate rejection, Banff score was II-III; control group met the diagnostic criteria of mild acute rejection, Banff score was 0-I (Figure 2, A3 and B3). At the end of 72 hours after operation, hematoxylin-eosin staining indicated the hepatic tissue of experiment group had a severe rejection and Banff score was III; control group presented the mild rejection, and Banff score was 0-I; rejection of the experimental group was severer than in the control group. Significant difference in Banff score was detected between the two groups (P < 0.05) (Figure 5, A4 and B4). Complications All receptors were noted, venous thrombosis and biliary complications. No severe complications of abdominal abscess. Two receptors were visible in hepatic artery thrombosis patients, but not directly killing receptors. "
| [1] Klein KB, Stafinski TD, Menon D. Predicting survival after liver transplantation based on pre-transplant MELD score: a systematic review of the literature. PLoS One. 2013;8(12): e80661.
[2] Alex Bishop G, Bertolino PD, Bowen DG, et al. Tolerance in liver transplantation. Best Pract Res Clin Gastroenterol. 2012;26(1):73-84.
[3] Xie XJ, Ye YF, Zhou L, et al. Th17 promotes acute rejection following liver transplantation in rats. J Zhejiang Univ Sci B. 2010;11(11):819-827.
[4] Stefanidis C, Callebaut G, Ngatchou W, et al. The role of biventricular assistance in primary graft failure after heart transplantation. Hellenic J Cardiol. 2012;53(2):160-162.
[5] Casas-Melley AT, Falkenstein KP, Flynn LM, et al. Improvement in renal function and rejection control in pediatric liver transplant recipients with the introduction of sirolimus. Pediatr Transplant. 2004,8(4):362-366.
[6] Dong JY, Yin H, Li RD, et al. The relationship between adenosine triphosphate within CD4(+) T lymphocytes and acute rejection after liver transplantation. Clin Transplant. 2011;25(3):E292-E296.
[7] Hashimoto K, Miller C, Hirose K, et al. Measurement of CD4+ T-cell function in predicting allograft rejection and recurrent hepatitis C after liver transplantation. Clin Transplant. 2010;24(5):701-708.
[8] Sun Y, Yin S, Xie H, et al. Immunophenotypic shift of memory CD8 T cells identifies the changes of immune status in the patients after liver transplantation. Scand J Clin Lab Invest. 2009;69(7):789-796.
[9] Gerlach UA, Vogt K, Schlickeiser S, et al. Elevation of CD4+ differentiated memory T cells is associated with acute cellular and antibody-mediated rejection after liver transplantation. Transplantation. 2013,95(12):1512-1520.
[10] Kubota N, Sugitani M, Takano S, et al. Correlation between acute rejection severity and CD8-positive T cells in living related liver transplantation. Transpl Immunol. 2006,16(1): 60-64.
[11] The Ministry of Science and Technology of the People’s Republic of China. Guidance Suggestions for the Care and Use of Laboratory Animals. 2006-09-30.
[12] Rang JH, Zhang SN, Liu J, et al. Improvements of Orthotopic Liver Transplantation Model in Rhesus Monkey. Zhongguo Puwaijichu yu Linchuang Zazhi. 2010;17(7):694-698.
[13] Rang JH, Li Z, Liu J, et al. Construction of stable rhesus monkey orthotopic liver transplantation models using two-cuff technique. Zhongguo Zuzhi Gongcheng Yanjiun yu Linchuang Kangfu. 2011;15(5):762-768.
[14] Solez K. History of the Banff classification of allograft pathology as it approaches its 20th year. Curr Opin Organ Transplant. 2010;15(1):49-51.
[15] Demetris A, Adams D, Bellamy C, et al. Update of the international banff schema for liver allograft rejection: working recommendations for the histopathologic staging and reporting of chronic rejection. An International Panel. Hepatology. 2000;31(3):792-799.
[16] Alcher A, Hayden LM, Brady WA, et al. Characterization of human inducible costinulator ligand expression and function. J Immunol. 2000;164(9):4689-4696.
[17] Coyle AJ, Gutierrez-Ramos JC. The expanding B7 superfamily increasing complexity in costinulatory signals regulating T cell function. Nat Immunol. 2001;2(3):203-209.
[18] Tafuri A, Shahinian A, Bladt F, et al. ICOS is essential for effective T-helper-cell responses. Nature. 2001;409(6816): 105-109.
[19] Coyle AJ, Lehar S, Lloyd C, et al. The CD28-related molecule ICOS is required for effective T cell-dependent immune responses. Immunity. 2000;13(1):95-105.
[20] Engelhard VH. Structure of peptides associated with class I and class II MHC molecules. Annu Rev Immunol. 1994; 12:181-207.
[21] MacNamara A, Kadolsky U, Bangham CR, et al. T-cell epitope prediction: rescaling can mask biological variation between MHC molecules. PLoS Comput Biol. 2009;5(3): e1000327.
[22] Stranzl T, Larsen MV, Lundegaard C, et al. NetCTLpan: pan-specific MHC class I pathway epitope predictions. Immunogenetics. 2010;62(6):357-368.
[23] Ossevoort MA, De Bruijn ML, Van Veen KJ, et al. Peptide specificity of alloreactive CD4 positive T lymphocytes directed against a major histocompatibility complex class-I disparity. Transplantation. 1996;62(10):1485-1491.
[24] Kovalik JP, Singh N, Mendiratta SK, et al. The alloreactive and self-restricted CD4+ T cell response directed against a single MHC class II/peptide combination. J Immunol. 2000; 165(3):1285-1293.
[25] Mendiratta SK, Kovalik JP, Hong S, et al. Peptide dependency of alloreactive CD4+ T cell responses. Int Immunol. 1999;11(3):351-360.
[26] Casares S, Bona CA, Brumeanu TD. Modulation of CD4 T cell function by soluble MHC II-peptide chimeras. Int Rev Immunol. 2001;20(5):547-573.
[27] Sadegh-Nasseri S, Dalai SK, Korb Ferris LC, et al. Suboptimal engagement of the T-cell receptor by a variety of peptide-MHC ligands triggers T-cell anergy. Immunology. 2010;129(1):1-7.
[28] Mirshahidi S, Huang CT, Sadegh-nasseri S. Anergy in peripheral memory CD4(+) T cells induced by low avidity engagement of T cell receptor. J Exp Med. 2001;194(6): 719-731.
[29] Choi S, Schwartz RH. Molecular mechanisms for adaptive tolerance and other T cell anergy models. Semin Immunol. 2007;19(3):140-152.
[30] Schwartz RH. T cell anergy. Annu Rev Immunol. 2003;21: 305-334.
[31] Coutinho A, Caramalho I, Seixas E, et al. Thymic commitment of regulatory T cells is a pathway of TCR-dependent selection that isolates repertoires undergoing positive or negative selection. Curr Top Microbiol Immunol. 2005;293:43-71.
[32] Chen W, Murphy B, Waaga AM, et al. Mechanisms of indirect allorecognition in graft rejection: class II MHC allopeptide-specific T cell clones transfer delayed-type hypersensitivity responses in vivo. Transplantation. 1996; 62(6):705-710.
[33] Slavcev A. Mechanisms of allorecognition and organ transplant rejection. Ann Transplant. 2001;6(4):5-8.
[34] Fujio K, Okamura T, Sumitomo S, et al. The functions of CD4+CD25-LAG3+ regulatory T cells and Egr2 in the regulation of autoimmunity. Nihon Rinsho Meneki Gakkai Kaishi. 2014;37(2):69-73.
[35] Wilczynski JR, Radwan M, Kalinka J. The characterization and role of regulatory T cells in immune reactions. Front Biosci. 2008;13:2266-2274.
[36] Shiao SL, Mcniff JM, Pober JS. Memory T cells and their costimulators in human allograft injury. J Immunol. 2005; 175(8):4886-4896.
[37] Vu MD, Clarkson MR, Yagita H, et al. Critical, but conditional, role of OX40 in memory T cell-mediated rejection. J Immunol. 2006;176(3):1394-1401.
[38] Vu MD, Amanullah F, Li Y, et al. Different costimulatory and growth factor requirements for CD4+ and CD8+ T cell-mediated rejection. J Immunol. 2004;173(1):214-221.
[39] Porrett PM, Lee MK 4th, Lian MM, et al. A direct comparison of rejection by CD8 and CD4 T cells in a transgenic model of allotransplantation. Arch Immunol Ther Exp (Warsz). 2008;56(3):193-200.
[40] Youssef AR, Otley C, Mathieson PW, et al. Role of CD4+ and CD8+ T cells in murine skin and heart allograft rejection across different antigenic desparities. Transpl Immunol. 2004;13(4):297-304. |
| [1] | Jiang Xin, Qiao Liangwei, Sun Dong, Li Ming, Fang Jun, Qu Qingshan. Expression of long chain non-coding RNA PGM5-AS1 in serum of renal transplant patients and its regulation of human glomerular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 741-745. |
| [2] | Li Liqiang, Jiao Longxing, Zhang Wu, Yan Wentao, Li Jian, Li Minghao. Effect of immature dendritic cells derived from bone marrow on rejection of orthotopic liver transplantation in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2025-2029. |
| [3] | Yang Feng, Chang Lipu, Huang Changshan, Gong Xiaoguang, Chang Shunwu. Macrolide antibiotics protects against ischemia-reperfusion injury after liver transplantation in rats [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(26): 4176-4182. |
| [4] | Liu Ting, Yang Tingting, Ma Xiaona, Ma Haibin, Jin Yiran, Liang Xueyun. Immunoregulation of allograft rejection: a role played by human CD200+ sub-population from human placenta-derived mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(13): 2068-2073. |
| [5] | Liu Mengyuan1, Fang Fang2. Risk factors for multi-drug resistant organisms infection after liver transplantation [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(7): 1109-1114. |
| [6] | Gao Hongqiang, Liu Jing, Li Zhiqiang, Wang Hailei, Zhao Xiongqi, Zhang Shengning, Ran Jianghua, Li Li . Ulinastatin improves rat liver metabolism after reduced-size liver transplantation [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(3): 435-440. |
| [7] | Cai Qiucheng, Fan Hongkai, Xiong Rihui, Jiang Yi. Intravenous administration of bone marrow mesenchymal stem cells protects liver function following fatty liver transplantation from donors after cardiac death [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(17): 2625-2629. |
| [8] | Zeng Rong-hua, Zhou Lu, Ouyang Hou-gan, Peng Shan, Gao Shu-liang, Wang Jian-min, Wu Hui-ting, Ouyang Yan-chu, Cui Tian-tian. Effect of acupuncture at Zusanli on T lymphocyte subsets in mesenteric lymph nodes of spleen deficiency syndrome rats [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(4): 576-581. |
| [9] | Zhao Ying-peng, Li Li, Chen Gang, Bai Jian-hua, Liu Qi-yu. Reduced-size liver transplantation with fatty liver donors in a rat model [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(4): 582-586. |
| [10] | Ding Chen1, Jiang Yi2, Pan Fan2, Cai Qiucheng2. Lipid metabolism after autologous orthotopic liver transplantation in rat models of fatty liver [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(36): 5821-5827. |
| [11] | Gong Yu-bo1, Liu Yong2, Zhao Hong-wei1, Xu Qian-qian1, Guo Hui-ling1 . Expression levels of Foxp3 and indoleamine 2,3-dioxygenase in mouse corneal allograft rejection [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(28): 4513-4517. |
| [12] | Liu Li-ye1, Zhao De-fang2, Gao Fei1, Zhang Tong2, Dong Qin2. Schisandra extract regulates Th17 cells and regulatory T cell imbalance in dogs undergoing liver transplantation [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(20): 3213-3217. |
| [13] | Tian Ying, Wu Jie, Wang Shuang-yong. Polyethylene glycol: an expert of cellular camouflage confusing the immune system [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(10): 1625-1633. |
| [14] | Liu Feng, Liang Zai-ming. Titanium and nano-hydroxyapatite for bone defect repair in rats: effects on immune regulation [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(10): 1506-1510. |
| [15] | Chen Jiang-bo, Pan Xi-min, Chen Ying-ming, Wu Zhi-qiang, Meng Zhong-meng, Chen Li-qiang, Zhuang Wen-quan. Magnetic resonance T2 mapping and T1ρ imaging of adult rhesus monkeys with lumbar intervertebral disc degeneration in free-range population [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(3): 418-422. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||